Antibody-drug conjugates (ADCs) combine the specificity of monoclonal antibodies with the potency of highly cytotoxic drugs, effectively delivering the drug payload to tumor sites and potentially reducing the severity of side effects. They are often referred to as the "magic bullets" of cancer therapy.
ADCs are increasingly being used in combination with other drugs, including for first-line cancer treatment. As the technology for producing these complex therapies matures, more ADC drugs are being approved or entering late-stage clinical trials. The diversification of antigen targets and biologically active payloads is rapidly expanding the range of tumor indications for ADCs. Additionally, new forms of carrier proteins and warheads targeting the tumor microenvironment hold promise for improving the tumor distribution or activation of ADCs, thereby enhancing their anticancer activity against refractory tumors. However, toxicity remains a key issue in the development of these drugs. Better understanding and management of ADC-related toxicities are crucial for their further optimization.
On June 12th, a review published in Nature Reviews Drug Discovery provided a detailed overview of the latest advances and challenges in ADC development for cancer therapy. Selected key points from the article are shared below.

Overview of ADCs on the Market
ADCs are complex therapeutic drugs composed of three key components: antibodies, linkers, and payloads (Figure 1). Optimizing each of these components can generate improved ADCs.
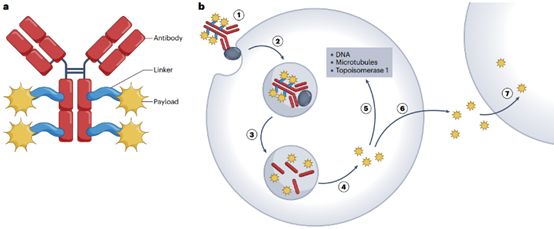
Figure 1 | Structure and mechanism of action of traditional ADCs
Currently, there are over a dozen ADCs approved for marketing worldwide. Among them, six target different antigens in hematologic malignancies: CD33, CD30, CD22, CD79b, BCMA, and CD19. Additionally, seven target different antigens expressed in solid tumors: HER2, nectin-4, TROP2, tissue factor (TF), and folate receptor alpha (FRα).
Solid tumors offer broad opportunities for ADC development as they are more common than hematologic malignancies, have limited treatment options in advanced or metastatic cases, and currently have few curative options through immunotherapy. In this regard, the approval of nectin-4-targeting enfortumab vedotin (Padcev) for locally advanced or metastatic urothelial carcinoma in 2019, and TROP2-targeting sacituzumab govitecan (Trodelvy) for triple-negative breast cancer in 2020, are significant as they provide new options for select, limited-disease populations.
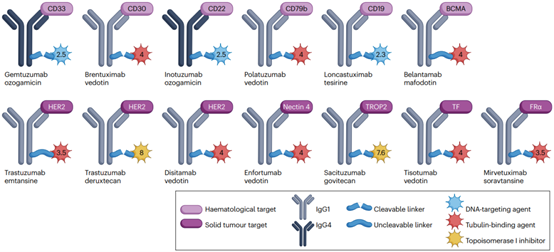
Figure 2 | Key characteristics of approved ADCs
Furthermore, as of December 2022, the ADC candidate trastuzumab duocarmazine is under regulatory review in the United States (with a PDUFA date of May 12, 2023). Two other ADCs, datopotamab deruxtecan and tusamitamab ravtansine, may have their marketing applications submitted in 2023.
Interestingly, approved ADC drugs are all based on cysteine conjugation (DAR 4-8) or random lysine conjugation (DAR 2/3-5). While site-specific conjugation has shown promising results in vitro and in vivo, it has not yet been successful in the clinical setting. Many of these ADCs have failed in clinical trials from Phase I to II, with only two out of 21 ADCs currently in late-stage trials (ARX788 from Ambrx and pivekimabsunirine/IMGN632 from ImmunoGen) being based on site-specific conjugation methods. Additionally, the majority of approved ADCs (11/13) as well as those in current late-stage clinical development (19/21) have cleavable linkers and non-polar payloads, allowing bystander killing effects.
Addressing the Limitations of ADCs
Like most drugs, the discontinuation of ADC development is often due to lack of efficacy, safety concerns, commercial considerations, or a combination of these factors. Since 2000, among the 97 ADCs that entered clinical trials and were terminated: the majority (81; 84%) were terminated in Phase I or I/II, with only 12 and 4 terminated in Phase II and III, respectively; most (67%) contained microtubule-binding payloads, 24% contained DNA-targeting agents (including two calicheamicin payloads and 21 PBD derivatives), and 3% contained topoisomerase 1 inhibitors; many (80%) targeted tumor antigens that were not representative of approved therapies, while 18 targeted validated targets; furthermore, 32 ADCs were terminated due to lack of efficacy, 32 due to safety issues, and 29 due to commercial considerations; several were developed for hematologic malignancies, while the majority were developed for solid tumors.
What lessons can be learned from these terminated projects? Insufficient anti-tumor activity at the maximum tolerated dose appears to be a major reason for termination. Additionally, unacceptable toxicity remains a significant obstacle in the development of novel ADC drugs. Better prediction of anticipated severe adverse events would be another approach to reduce premature termination of drug development. For example, the ADC HTK288 targeting cadherin-6 was associated with unexpected central nervous system toxicity, while the ADC LOP628 targeting the tyrosine kinase receptor KIT was associated with unexpected severe hypersensitivity reactions. As demonstrated in the development of cisplatin and paclitaxel in the past, managing the toxicity of a new compound is often a long-term endeavor.
In summary, finding the right combination of target antigen, active linker-payload, appropriate DAR value, and suitable tumor indication is challenging. With the rapid growth of validated target antigens and the diversification of payloads, it is expected that further development is needed for some terminated ADCs targeting specific antigens.
1. Diversification of Payloads
Prior to the approval of trastuzumab deruxtecan in 2019, approved ADCs could be categorized into two major classes of payloads: microtubule-binding agents and DNA-targeting agents. Now, many other drug molecules are being evaluated as potential payloads.
Auristatin derivatives interfere with microtubule polymerization dynamics, leading to disruption of mitotic spindle formation, mitotic arrest, and subsequent cell death, making them potent payloads. ADCs containing Auristatin are currently the largest family of ADCs (Figure 3).
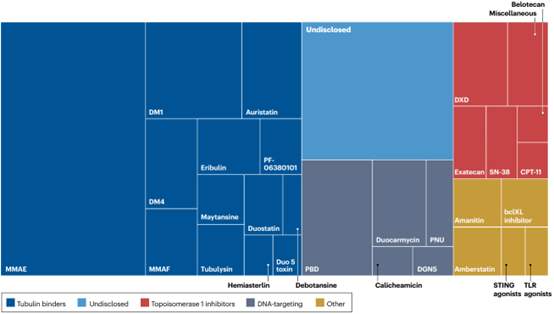
Figure 3 | Diversification of payloads in ADC pipelines
The second major representative payload class is DNA-targeting agents, which chemically modify DNA to prevent cell replication. Calicheamicin is a potent DNA-damaging agent that induces double-strand DNA (dsDNA) breaks through a free radical mechanism and is used as the payload in two approved ADCs. PBD dimers are alkylating agents that crosslink dsDNA and are among the most potent identified cytotoxic agents.
Until recently, most ADCs in development used highly potent cytotoxins as "warheads". This was partly due to observations that high DAR values were associated with less favorable pharmacokinetic properties, thus favoring the use of more potent payloads that generate effective ADCs at DAR。
Given that not all types of tumors are sensitive to a given payload, payload diversification is crucial for expanding the indications of ADCs. Over the past two decades, we have witnessed the successful development of ADCs, with powerful members in this family. Regarding payload development, three observations can be made. Firstly, not all commonly used cytotoxic agent families have been successfully used as ADC payloads. In particular, attempts to develop nucleoside analogs and antimetabolites as payloads have failed. Secondly, there are currently no approved ADCs that contain payloads with cytotoxic mechanisms distinct from traditional chemotherapy. Lastly, a large number of novel drugs with primary mechanisms of action, including kinase inhibitors and molecules targeting various processes within tumor cells, have failed in clinical trials due to poor safety. However, some of these drugs may be potential candidates as ADC payloads. Heidelberg Pharma has brought an example of this type of development into clinical practice, using α-Amanitin derivatives as novel ADC payloads.
Given the complexity of ADCs, payload diversification may be seen as a risky endeavor. If the payload combines with a validated target (such as HER2), the results can be highly promising, as seen with trastuzumab emtansine. However, it is difficult to estimate how many second or later-generation ADCs will successfully be developed for a given target. On the other hand, exploring novel payloads associated with unvalidated targets increases the risk of failure and complicates the assessment of its underlying reasons.
2.Importance of Toxicity
Based on the side effects typically associated with the considered payload type, toxicity associated with ADC administration can be categorized as "expected" and "unexpected."
Expected toxicity includes, for example, MMAE-induced peripheral neuropathy, which is a typical side effect of tubulin-binding agents. Myelosuppression is a common complication of most cytotoxic chemotherapy drugs, especially DNA-targeting agents.
Unexpected toxicity: For example, while ADCs based on MMAE are typically not associated with ocular toxicity, ADCs using MMAF as a approved payload (such as belantamab mafodotin) have been associated with corneal toxicity, with up to 72% of patients showing epithelial changes. Additionally, as ADCs are increasingly incorporated into combination regimens, other unexpected toxicities may be observed. In Hodgkin lymphoma patients, the combination of brentuximab vedotin (BV) with a standard care regimen containing bleomycin resulted in severe pulmonary toxicity in 44% of patients, while it was absent in the group without bleomycin. The underlying mechanisms for these diverse toxicities are not fully understood and may involve Fc-mediated ADC internalization, release of payload in normal tissues, off-tumor expression of the target antigen, non-specific uptake of ADCs through phagocytosis or similar cellular processes, enzyme-mediated release of payload in systemic or specific normal tissue environments, or Fc-mediated inflammatory effects in the context of payload-mediated tissue damage.
Given that ADCs are increasingly being used as first-line or adjuvant therapies, the long-term safety and reversibility of side effects become increasingly important. One important concern is the potential mutagenic effects of certain payloads, particularly DNA-targeting agents. Peripheral neuropathy may be irreversible in some patients, underscoring the necessity of adaptive dosing and close monitoring in at-risk patients. Another key issue is determining specific dosing regimens for certain patients based on variables such as age, gender, type and quantity of prior treatments, comorbidities, or genetic characteristics.
3.Overcoming Resistance
Considering the series of steps required for successful cytotoxic action of ADCs, the potential mechanisms of ADC resistance may be complex. Resistance to ADCs can be observed in cases where antigen binding and/or antibody/antigen internalization are reduced, intracellular concentrations of the payload are decreased, changes occur in the target of the payload, or alterations in apoptosis mechanisms.
Several therapeutic interventions can enhance the efficacy of ADCs in preclinical models. One possibility is to enhance ADC internalization, the mechanisms of which are still not fully understood. Overexpression of caveolin-1, associated with lipid rafts, increased the internalization of T-DM1. In cases where resistance has already been established, the use of a second type of payload can be considered.
Antigen expression heterogeneity is a classic mechanism for the failure of antibody-based therapies. Preclinical models have shown that the distribution of antibodies in tumors depends on the expression of the target antigen. The bystander effect provided by most ADCs, including the release of unconjugated payload in the tumor microenvironment and its potential uptake by neighboring tumor cells independent of their antigen profile, serves as a powerful means to counteract antigen heterogeneity within tumors, as the payload can reach tumor cells with low levels of the target antigen.
Furthermore, the expected mode of administration of ADCs to patients also strongly influences the occurrence of resistance. In early trials, ADCs were primarily administered as monotherapy, which promoted the selection of resistant tumor populations. Currently, many combination regimens, including traditional cytotoxic chemotherapy and other targeted agents, are being explored in clinical practice. The treatment sequence may also be an important parameter.
4.Combination Therapy
Brentuximab vedotin (BV) has been used in combination with over 80 different types of regimens, including cytotoxic chemotherapy and immune checkpoint inhibitors. The combination of BV with immune checkpoint inhibitors shows great promise. After demonstrating the efficacy of single-agent pembrolizumab in relapsed Hodgkin lymphoma patients and its superior activity compared to BV in this indication, some studies have explored the combination of BV with anti-PD-1/PD-L1 or anti-CTLA-4 agents. In relapsed/refractory Hodgkin lymphoma patients, the combination therapy of BV with nivolumab induces an 82% overall response rate (ORR), including 61% complete response (CR).
T-DM1 has also been explored in combination regimens. Adding capecitabine to T-DM1 did not improve ORR but led to more adverse events. T-DM1 in combination with pertuzumab showed contrasting results. In the MARIANNE study, patients with advanced breast cancer treated with T-DM1 in combination with pertuzumab had similar overall survival but better quality of life compared to those receiving pertuzumab and paclitaxel. T-DM1 has also been combined with immune checkpoint inhibitors, with support from clinical and preclinical data.
Overall, these studies suggest that carefully selected combinations of ADCs with other drugs may be superior to antibody-based therapies in terms of patient outcomes and safety. Safety is a key concern in combination design, especially for frail or comorbid patients. Overlapping or unexpected toxicities need to be closely monitored and adjusted based on individual patient circumstances. Future research will need to determine which patient subgroups benefit most from these combinations.
The Future of ADC Development
ADCs have established a solid position in the oncology armamentarium. There are over 1500 clinical studies listed on clinicaltrials.gov involving ADCs, with an increasing number of drugs entering clinical trials (Figure 4). It can be expected that the granted approvals for ADCs will diversify significantly, as well as their indications in various diseases.
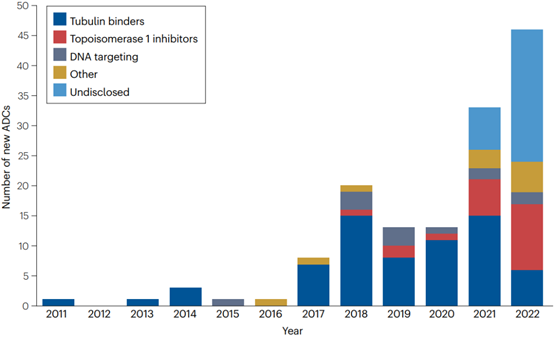
Figure 4 | Number of new ADCs entering clinical trials between 2012 and 2022. The number of new ADCs entering clinical evaluation has rapidly increased in recent years. Since 2021, the proportion of ADCs containing topoisomerase 1 inhibitors has increased. The proportion of undisclosed payloads is also increasing (48% in 2022).
Currently approved ADCs are almost evenly distributed between hematologic malignancies and solid tumors. The challenges in developing ADCs for solid tumors may stem from specific features, including poor diffusion, intrinsic resistance to cytotoxic drugs, and decreased mitotic fraction. Enhanced tumor penetration may enhance ADC activity in solid tumor indications, either by using smaller formats or employing probody-mediated tumor activation. Several promising targets are currently being evaluated in the clinic for solid tumors, such as ROR1, HER3, CEACAM5, MET, and NaPi2b, and numerous tumor-associated antigens are being evaluated as potential targets for ADC-mediated drug delivery.
The expected developments in ADCs will include new target antigens, payloads with novel mechanisms of action, new linker technologies that offer better therapeutic indices, and novel antibody and carrier formats.
1.Inducing Immunogenic Cell Death
There is an increasing focus on the immunostimulatory properties of ADCs. In addition to the binding of immunostimulatory agents themselves, as in immune-activating antibody-drug conjugates (iADCs), ADCs can induce immunogenic cell death (ICD), thereby promoting anti-tumor immune responses. The induction of ICD by ADCs may be a contributing factor to their effectiveness in combination with immune checkpoint inhibitors, particularly in immune-rich diseases like Hodgkin lymphoma. Belantamab mafodotin induces ICD in vivo and activates dendritic cells in immunocompetent mouse models. ADCs based on anthracycline drugs targeting HER2 have also been shown to induce ICD and immunogenic memory. The ability of ADC payloads to induce ICD may vary, and further studies will help determine their potential as immunostimulatory agents.
2.Targeting Extracellular Antigens
The initial paradigm for anti-cancer ADCs was based on intracellular release of cytotoxic payloads, which relied on internalization. The bystander effect and the ability of the payload to diffuse within the tumor depend on the physicochemical properties and potency of the payload. An exception to this rule is being investigated with non-internalizing antibodies targeting components of the tumor microenvironment. For example, PNU-conjugated antibodies targeting the splice isoforms of tenascin C have shown complete remission in preclinical models. Galectin-3 binding protein (LGALS3BP), preferentially secreted by tumor cells, has been explored as an extracellular target for ADCs. Other potential extracellular targets for ADC-mediated drug delivery can be identified through high-throughput computational approaches.
While the mechanisms of action for these formulations are highly innovative, they face specific challenges, including relative expression of the target antigen in normal tissues versus tumor tissues, sufficient release of payload in the environment, and effective penetration of the payload within tumor cells. However, establishing approaches that explicitly guide ADCs toward extracellular targets build on the concept that diffusible bystander payloads released extracellularly may be an underappreciated component of many mechanisms by which ADCs target solid tumors.
3.Eliminating Immunosuppressive Tumor Microenvironment
A third alternative to targeting tumor cells themselves or extracellular antigens is the depletion of cell populations that impact therapeutic outcomes. Saha et al. demonstrated successful myeloablation with an anti-CD45 ADC in mice receiving allogeneic hematopoietic stem cell transplantation, indicating the potential to spare patients from whole-body irradiation or exposure to potent alkylating agents. Recently, a marrow-ablative CD117 alpha-amanitin ADC showed good tolerability in phase I/II studies. As our understanding of immunosuppressive cells in the tumor microenvironment increases, ADCs may be developed to deplete specific cell populations such as regulatory T cells (Tregs), M2 macrophages, or myeloid-derived suppressor cells.
4.Beyond Standard ADC Formats
While the development of ADCs has largely relied on standard single-specificity antibodies for targeting, the industry is exploring alternatives such as probody-drug conjugates (PDCs) and biparatopic or bispecific ADCs to enhance tumor specificity and reduce toxicity to healthy tissues. PDCs are masked, proteolytically cleavable prodrugs designed to provide therapeutic effects within tumors by leveraging the dysregulated tumor protease activity in the tumor microenvironment. Probody masking peptides prevent binding to targets in healthy tissues.
The rapid development of bispecific antibody technology has provided more options for antibody formats used in ADCs. Combining payloads on bispecific antibodies to generate bispecific ADCs with improved specificity and/or internalization is a new area of research that holds promise for overcoming existing limitations such as ADC internalization, toxicity, and resistance. Nine bispecific ADCs have entered phase I clinical trials, with one discontinued (MEDI4276). Among them, four are biparatopic targeting two different epitopes on the same target, and five are targeting two different tumor-associated antigens.
Outlook
ADCs have made a significant impact on clinical oncology. Several ADCs have experienced rapid sales growth, with three products generating over 1 billion Euros in sales in 2022 (BV, T-DM1, and T-DXd), confirming the broad clinical utility of ADCs.
One major characteristic driving ADC development is the significantly improved therapeutic index compared to non-conjugated (i.e., traditional) cytotoxic drugs. However, this assumption has been questioned recently in terms of MTD values and expanding the therapeutic window. Therefore, the extent to which ADCs will replace traditional cytotoxic chemotherapy remains to be determined.
Current limitations of ADCs include their high cost and lack of oral administration. Furthermore, ADCs are unlikely to be administered subcutaneously, while an increasing number of naked antibodies are being marketed as subcutaneous formulations. However, ADCs have established a robust position in the landscape of anticancer therapeutics, even though the development of these drugs is more complex than that of naked antibodies. It is expected that the number of approved ADCs will significantly increase in the coming years to meet the growing unmet medical needs in both common and rare diseases.
References:
Dumontet, C., Laïnée, J. M., Senter, P., et al. Antibody-drug conjugates in oncology: The maturation of a breakthrough. Nature Reviews Drug Discovery (2023).
Bailu,The article on the WeChat official account of Medicine Rubik's Cube is deep and good! ADC treatment for cancer, how to take the future path
Note: Figures in the text are sourced from the Nature Reviews Drug Discovery article.